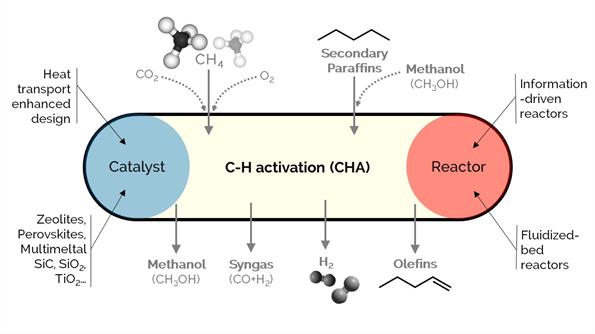
Methane and light alkanes are surplus species and by-products with relatively poor economic interest. Our goal is to activate C–H σ-bond to produce hydrogen, olefins, carbon monoxide, and carbon nanofibers, following different process strategies such as oxidative coupling (for olefins), CO2 dry reforming (for syngas), cracking or catalytic decomposition (for hydrogen-free of COx and sequestrated carbon nanotubes/nanofibers), cracking/co-cracking with CO or methanol. We work on developing, synthesizing, characterizing, and testing innovative catalysts with a twist of reaction engineering concepts, looking at multi-scale implications.
We delve into the mechanistic insights of a series of in-house synthesized metal-supported heterogeneous catalysts by combining them with dynamic reactors and ab initio calculations. We explore catalysts with promoted lifetime, activity, selectivity, and heat exchange.
We investigate novel reactor designs grounded on forced dynamic (operando) fluidized-bed reactors at high pressures to amplify the kinetic information and hydrogen.